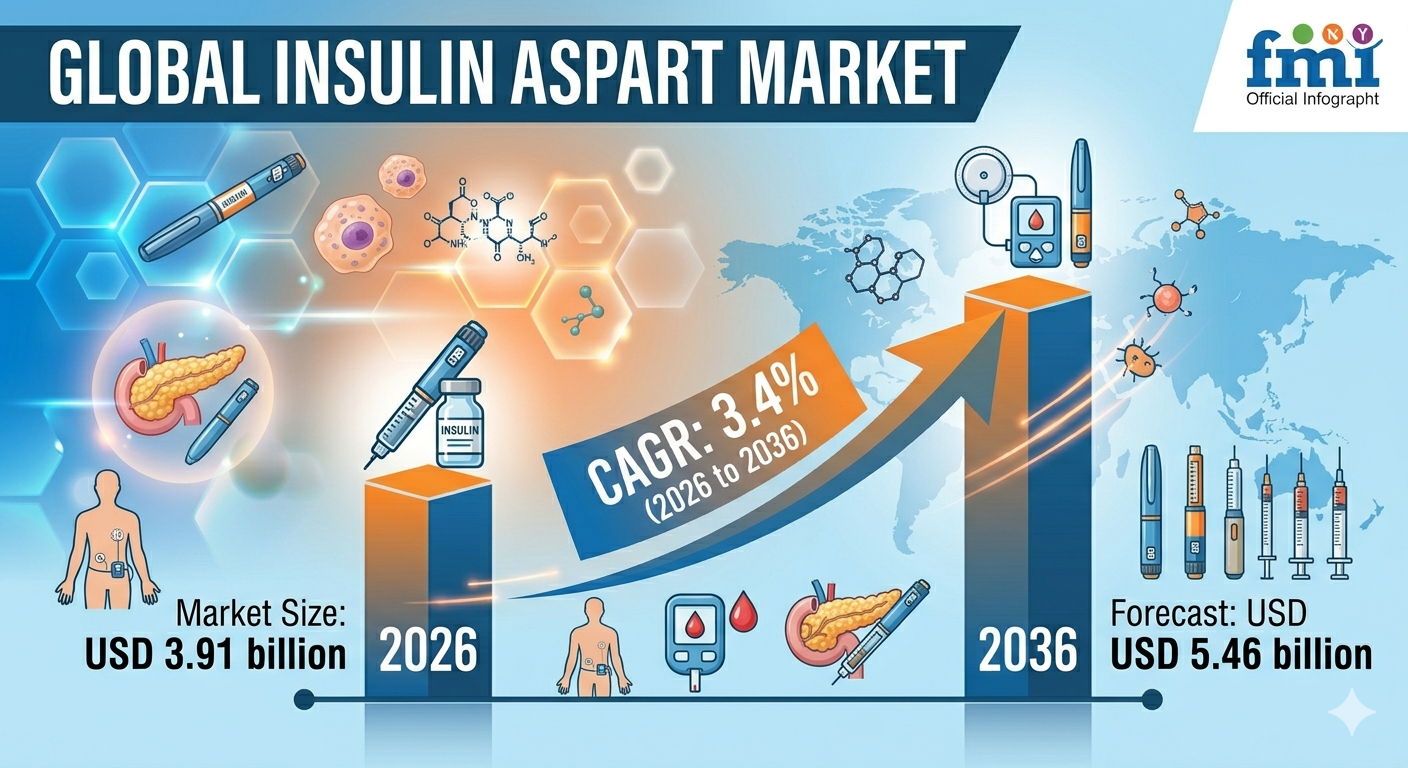
The insulin aspart market is estimated at USD 3.91 billion in 2026 and is projected to reach USD 5.46 billion by 2036, expanding at a CAGR of 3.4% during the forecast period.
NEWARK, DE / ACCESS Newswire / May 11, 2026 / According to the latest analysis by Future Market Insights, the Insulin Aspart Market is entering a new competitive phase shaped by biosimilar adoption, payer-driven switching strategies, and expanding diabetes prevalence worldwide. While rapid-acting insulin analogues remain central to intensive insulin therapy and mealtime glucose management, the market is increasingly transitioning from a brand-led growth model toward an access- and affordability-driven landscape.
Insulin aspart continues to play a critical role in type 1 diabetes care, insulin-treated type 2 diabetes, and pump-linked therapy due to its ability to provide rapid post-meal glucose lowering. However, recent biosimilar approvals are changing the market structure by giving healthcare systems, payers, and pharmacy benefit managers greater flexibility to reduce costs while maintaining treatment continuity.
The approval of rapid-acting insulin biosimilars such as Merilog and Kirsty in 2025 marks a major turning point for the category. As biosimilar competition expands, suppliers are increasingly competing on device convenience, pharmacy access, cold-chain reliability, and payer alignment rather than brand familiarity alone.
At the same time, the growing adoption of insulin pumps and hybrid closed-loop systems is sustaining long-term demand for rapid-acting insulin formulations. These advanced diabetes management technologies rely heavily on fast insulin action, reinforcing insulin aspart's importance across modern diabetes care pathways.
Detailed market forecasts, competitive benchmarking, and service trends: https://www.futuremarketinsights.com/reports/sample/rep-gb-33048
Quick Stats Snapshot
Market value (2025): USD 3.78 billion
Forecast (2036): USD 5.46 billion
CAGR (2026-2036): 3.4%
Incremental opportunity: USD 1.55 billion
Leading product type: Standard insulin aspart (62.0% share)
Dominant delivery format: Prefilled pens (57.0% share)
Largest indication segment: Type 1 diabetes (51.0% share)
Leading distribution channel: Retail pharmacies (54.0% share)
Fastest-growing markets: India (5.8% CAGR) and China (5.2% CAGR)
Market Size and Forecast: Rapid-Acting Insulin Maintains Strategic Relevance
The insulin aspart market is estimated at USD 3.91 billion in 2026 and is projected to reach USD 5.46 billion by 2036, expanding at a CAGR of 3.4% during the forecast period.
Despite increasing pricing pressure from biosimilars, insulin aspart remains deeply embedded in diabetes management because of its role in daily mealtime glucose control. Demand continues to be supported by repeat prescription use, intensive insulin therapy, and the expansion of automated insulin delivery technologies.
As diabetes prevalence continues to rise globally, particularly across emerging healthcare markets, long-term insulin demand is expected to remain structurally resilient.
Growth Drivers: Why Demand Remains Strong
Rising Global Diabetes Burden: According to the International Diabetes Federation, 589 million adults aged 20 to 79 were living with diabetes in 2024, with the number projected to reach 853 million by 2050. This expanding patient base continues to support long-term insulin demand globally.
Growth of Pump and Hybrid Closed-Loop Therapy: Insulin pumps and hybrid closed-loop systems increasingly depend on rapid-acting insulin analogues such as insulin aspart for automated insulin delivery and tighter glucose control. As adoption of these technologies grows, rapid insulin formats remain strategically important.
Biosimilar Expansion Improves Access: FDA approvals for Merilog and Kirsty in 2025 created new opportunities for lower-cost rapid-acting insulin access in the United States. Biosimilar availability is expected to improve affordability while increasing competitive pressure on legacy brands.
Preference for Convenient Delivery Formats: Prefilled pens continue gaining adoption because patients and caregivers prioritize simplified dosing, portability, and reduced administration errors compared with vial-based delivery systems.
Challenges: Areas of Market Pressure
Biosimilar-Driven Price Competition: The entry of interchangeable insulin aspart biosimilars is increasing formulary competition and limiting pricing flexibility for branded manufacturers.
Affordability Barriers in Emerging Markets: Although diabetes prevalence is rising sharply in lower-income regions, access to analogue insulin remains constrained by affordability, reimbursement gaps, and cold-chain limitations.
Payer and Formulary Pressure: Pharmacy benefit managers and national healthcare systems are increasingly emphasizing cost containment, driving substitution toward lower-cost insulin alternatives.
Slower Value Growth in Mature Markets: Markets such as the United States, Germany, and Japan already have high insulin penetration rates, limiting future growth primarily to switching activity and premium device formats.
Opportunities: Where Strategic Growth Is Emerging
Biosimilar Penetration Across Global Markets: Biosimilar insulin aspart products are expected to create broader treatment access while opening new growth opportunities for regional manufacturers and healthcare systems.
Expansion of Diabetes Care Infrastructure: Countries such as India and China are expanding formal diabetes treatment pathways, specialty endocrinology access, and chronic-care management systems.
Advanced Delivery Technologies: Growth in connected diabetes devices, smart pens, and automated insulin delivery systems is creating demand for highly reliable rapid-acting insulin formulations.
WHO Access Initiatives: The World Health Organization's expansion of the insulin analogue master file procedure is expected to improve global access to quality-assured insulin products in underserved markets.
Segmentation Insights: How the Market Is Structured
By Product Type: Standard insulin aspart is projected to account for 62.0% share in 2026 due to broad physician familiarity, long-standing clinical use, and established payer coverage.
By Delivery Format:Prefilled pens are estimated to represent 57.0% share as self-administration convenience continues shaping patient preference and chronic therapy adherence.
By Indication:Type 1 diabetes is expected to hold 51.0% share because intensive insulin therapy requires rapid-acting insulin coverage across daily meals.
By Distribution Channel: Retail pharmacies are projected to maintain 54.0% share due to chronic refill behavior and strong outpatient diabetes treatment demand.
Speak to Analyst: Customize insights for your business strategy: https://www.futuremarketinsights.com/customization-available/rep-gb-33048
Regional Analysis: Where Growth Is Concentrated
India (5.8% CAGR): India is projected to record the fastest market growth due to rising diabetes prevalence, expanding formal insulin access, and growing organized pharmacy infrastructure.
China (5.2% CAGR): China's insulin aspart market is benefiting from broader chronic disease management systems, urban diabetes care expansion, and increasing analogue insulin adoption.
South Korea (4.1% CAGR): South Korea's advanced diabetes care infrastructure and growing pump-linked therapy adoption continue supporting rapid-acting insulin demand.
United States (2.8% CAGR): Although the U.S. remains a high-value market, biosimilar approvals and payer pressure are moderating value growth.
United Kingdom and Germany: Mature reimbursement systems, strong procurement oversight, and established diabetes treatment pathways are sustaining steady but slower expansion.
Competitive Landscape: Biosimilars Reshape Market Dynamics
The insulin aspart market is increasingly defined by biosimilar competition, payer negotiations, and device-format differentiation.
Key participants include:
Novo Nordisk
Sanofi
Biocon Biologics
Viatris
Gan & Lee Pharmaceuticals
Tonghua Dongbao
Wockhardt
Novo Nordisk continues to hold the strongest reference-product position through NovoLog, NovoRapid, and Fiasp. However, biosimilar competition from Sanofi and Biocon Biologics is intensifying following FDA approvals for Merilog and Kirsty.
The next phase of competition is expected to focus on formulary access, patient switching behavior, device reliability, and cold-chain distribution performance.
Strategic Implications for Industry Stakeholders
For Manufacturers: Competitive success increasingly depends on pricing flexibility, payer relationships, and device convenience rather than legacy brand positioning alone.
For Payers: Biosimilar insulin aspart products provide new opportunities to manage diabetes treatment costs while maintaining therapeutic continuity.
For Healthcare Providers: Reliable patient education and switching management will become increasingly important as biosimilar adoption expands.
For Distribution and Pharmacy Networks: Cold-chain reliability and consistent pharmacy availability remain critical for long-term market penetration.
Future Outlook: Toward an Access-Driven Rapid Insulin Market
Over the next decade, the insulin aspart market is expected to evolve in three major directions:
Greater Biosimilar Penetration: Interchangeable biosimilars are expected to accelerate formulary switching and broaden treatment affordability across multiple regions.
Expansion of Automated Diabetes Care: Hybrid closed-loop systems and connected diabetes technologies will continue reinforcing demand for rapid-acting insulin formulations.
Increasing Focus on Emerging Markets: Long-term growth will increasingly shift toward India, China, and other developing healthcare systems where diabetes prevalence continues expanding rapidly.
Unlock 360° insights for strategic decision making and investment planning: https://www.futuremarketinsights.com/checkout/33048
Executive Takeaways
Insulin aspart remains central to mealtime glucose control across type 1 diabetes, insulin-treated type 2 diabetes, and pump-linked therapy.
Biosimilar approvals are transforming the competitive landscape by shifting market focus toward affordability and access.
Prefilled pens continue dominating delivery formats due to patient convenience and adherence advantages.
India and China are expected to deliver the strongest long-term growth as formal diabetes care infrastructure expands.
Competitive differentiation increasingly depends on payer alignment, pharmacy access, device reliability, and supply-chain consistency.
For organizations operating within diabetes therapeutics, the market outlook is becoming increasingly clear: the future of insulin aspart will be defined less by brand legacy and more by the ability to deliver affordable, reliable, and accessible rapid-acting insulin across an expanding global diabetes population.
Explore More Related Studies Published by FMI Research:
Insulin Pens Market: https://www.futuremarketinsights.com/reports/insulin-pens-market
Insulin Resistance Improvement Drug Market: https://www.futuremarketinsights.com/reports/insulin-resistance-improvement-drug-market
Insulin Biosimilar Market: https://www.futuremarketinsights.com/reports/insulin-biosimilars-market
Insulin Pumps Market: https://www.futuremarketinsights.com/reports/insulin-pumps-market
Insulin Management System Market: https://www.futuremarketinsights.com/reports/insulin-management-system-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com/
For Web : https://www.factmr.com/
SOURCE: Future Market Insights, Inc.
View the original press release on ACCESS Newswire





